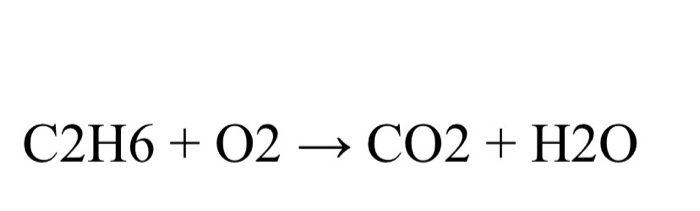Balance the following equation by oxidation number method. C6H6 + O2 → CO2 + H2 O - Sarthaks eConnect | Largest Online Education Community
When 10 g of CxHy is burned, 45.00 g of products are formed (CO2 and H2O combined), what is the empirical formula of the fuel (CxHy + O2→ CO2 + H2O)? - Quora

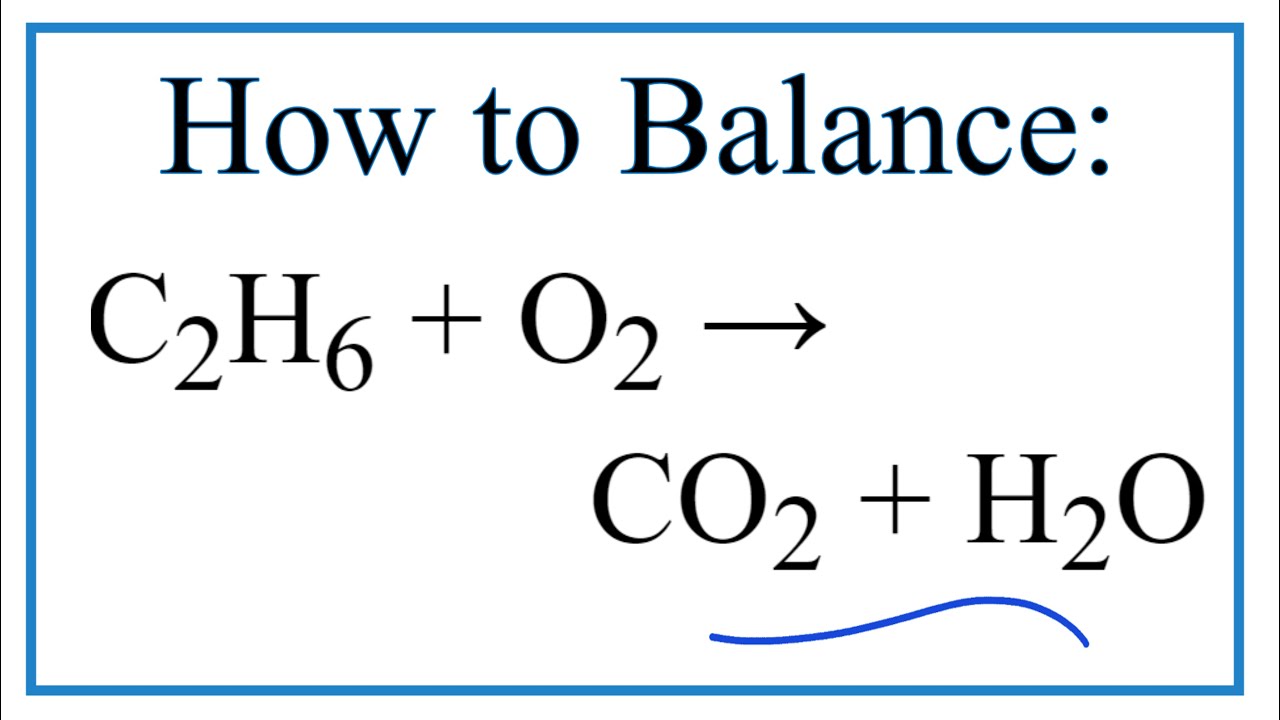
C2H6 +O2 =CO2 + H2O Balanced Equation C2H6+O2 combustion(Ethane) Balanced reaction and Equation - YouTube

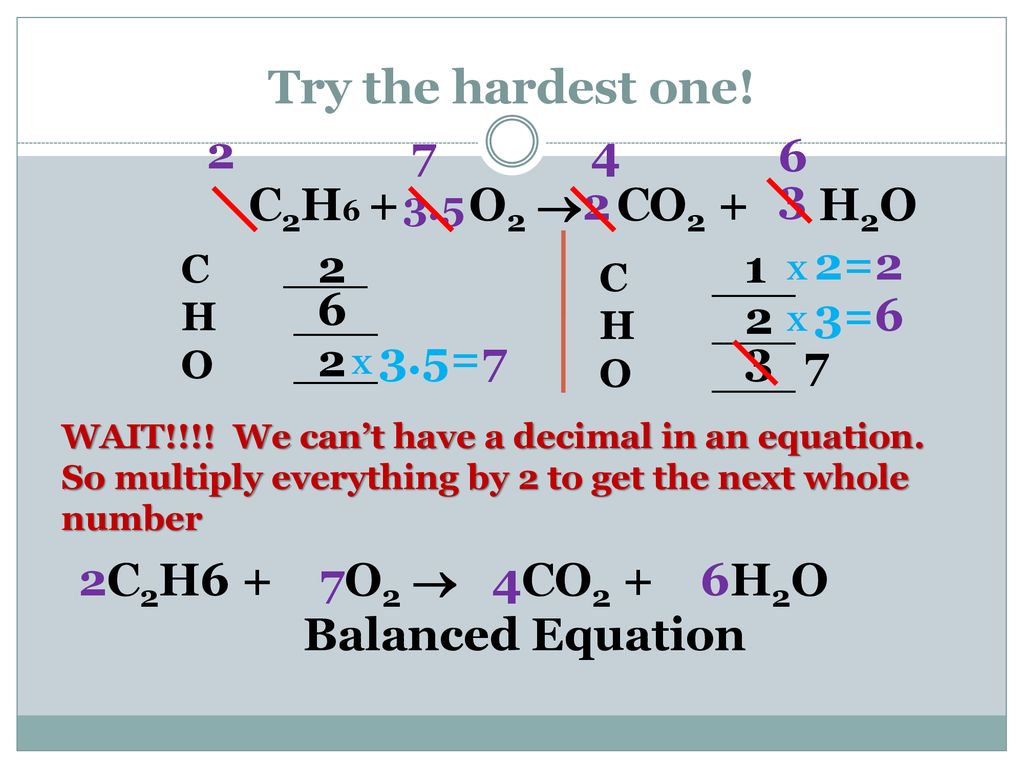
SOLVED: The following chemical equation shows the combustion of ethane. Balance the equation by filling in the coefficients. C2H6 + O2 → CO2 + H2O